In the optimal experimental conditions, the recoveries of yttrium oxide are about 95%, 55%, and 65% for CRT, lamps, and CRT/lamp mixture (called MIX) powders, respectively. Experimental results have shown that process conditions necessary to purify the solutions and recover yttrium strongly depend on composition of the leach liquor, in other words, whether the powder comes from treatment of CRTs or lamp. The process for treating these materials includes the following: (a) acid leaching, (b) purification of the leach liquors using sodium hydroxide and sodium sulfide, (c) precipitation of yttrium using oxalic acid, and (d) calcinations of oxalates for production of yttrium oxides. Abstract: In this paper, yttrium recovery from fluorescent powder of lamps and cathode ray tubes (CRTs) is described. • Economic simulation for the processes to recover yttrium from WEEE. • Recovery of yttrium from fluorescent powders. • Fluorescent powder of cathode ray rubes. Highlights: • Fluorescent powder of lamps. Recovery of yttrium from cathode ray tubes and lamps’ fluorescent powders: experimental results and economic simulation A first version of a mock-up is presented, and experimental results are presented and discussed. In the next part, the author addresses some theoretical aspects of corrections to be made for spot deflection, spot focussing, and spot brightness. The author describes a cathode ray tube, presents different methods to obtain a colour image (mask tube, electron penetration tube, and intensity change tube), discusses the choice of a cathode ray tube type, and describes its use in a display console. The objective of this research thesis is to study the possibility to obtain a colour image which can be used in cathode ray tube display console. Use of an electron penetration cathode ray tube in a colour display console The experimental results reviewed here include A-site and B-site segregations, lattice expansion, oxidation-state changes during cell operations and liquid-phase infiltration and coarsening of cathode to electrolyte backbone The epitaxial thin film model cathodes for XR, TXRF, and HRD measurements are made by pulse laser deposition and porous film cathodes for USAX measurements are made by screen printing technique. 
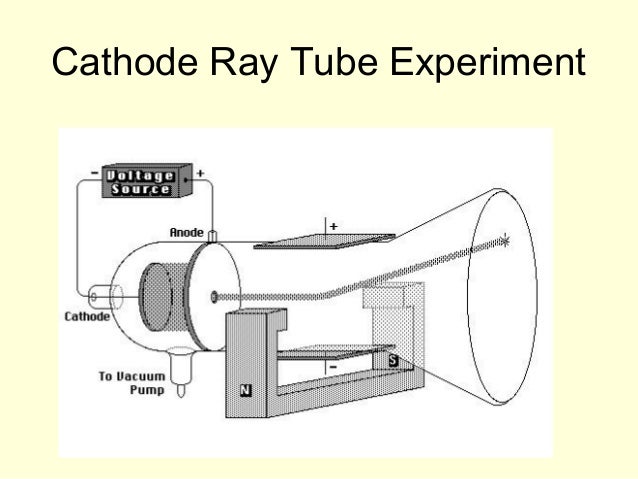
The X- ray techniques used include X- ray reflectivity (XR), total-reflection X- ray fluorescence (TXRF), high-resolution diffraction (HRD), ultra-small angle X- ray scattering (USAXS). Abstract: Synchrotron-based X- ray techniques have been used to study in situ the structural and chemical changes of film cathodes during half-cell operations. •Liquid-phase infiltration and coarsening processes of cathode materials into porous YSZ electrolyte backbone were monitored by USAXS. •The surface and subsurface of the LSM and LSCF films have different oxidation-states due to vacancy concentration changes. •Chemical lattice expansions show that oxygen- cathode interface is the primary source of rate-limiting processes. •A-site and B-site cations actively segregate or desegregate on the changes of temperature, pO 2, and electrochemical potential. Highlights: •Synchrotron X- rays are used to study in operando the structural and chemical changes of LSM and LSCF film cathodes during half-cell operations. International Nuclear Information System (INIS)įuoss, Paul Chang, Kee-Chul You, Hoydoo This was the beginning of further understanding leading to the atomic theory and structure that we know today.Ĭathode ray tube is a tube that contains a small amount of gasīetween two metallic plates.In situ X- ray studies of film cathodes for solid oxide fuel cells He won the 1906 Nobel prize for the discovery of electrons. He zapped atoms with electricity and observed that negatively charged particles were removed! He reasoned that atoms consisted of subatomic particles, electrons that were negatively charged particles. Thomson, a British physicist, conducted the cathode ray experiment. Previously, atoms were known to be indivisible, but in 1897, J. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
